The global Medical Devices industry has witnessed a rapid growth in the past decade fuelled by a growing Middle-Income class and rise in the Private Healthcare institutions in emerging markets. The 2008 global medical device market was estimated to be around $206.59 billion with an annual growth rate of nearly 16% and the latest figures would be much much higher.
Know more: Sage X3 for Healthcare Industry
However, the journey for this industry is not something smooth. Medical device manufacturer has to meet the challenging demands of the industry like regulatory agencies compliance, strict quality assurance, and short product life cycles etc. The manufacturer has to maintain efficient production activities along with compliance with guidelines laid by FDA as done by Pharmaceutical industry. Let alone the increased product complexity, time to market and competitive challenges.
Keeping in mind these challenges, Sage X3 ERP has been fine-tuned to meet these industry-specific requirements. Sage ERP X3 provides the advanced functionality required to remain competitive in the face of these difficult challenges.
Industry Specific Functionalities
Sage ERP X3 combines advanced manufacturing, distribution, warehousing, customer relationship management (CRM), and accounting functionality with application features that address the unique and specialized needs of your company, including:
- Full lot and serial number traceability.
- Electronic signatures with transaction audit traceability.
- Quality management.
- Sophisticated pricing and contract management.
- Sub-contracted operations and supply chain control.
- Post-sale service and support.
- Extensive device history.
- Advanced order fulfillment and inventory allocation procedures.
Compliance with Regulatory Agencies
Compliance with the government regulations requires hosts of reports. This calls for accessing and compiling the data required for such government reporting. This again entails expensive labor and time consumption. Sage ERP X3 also helps your company comply by providing a convenient and efficient way to gather and monitor relevant process information and eliminating manual procedures throughout the manufacturing process.
Sage X3 ERP Application Footprints
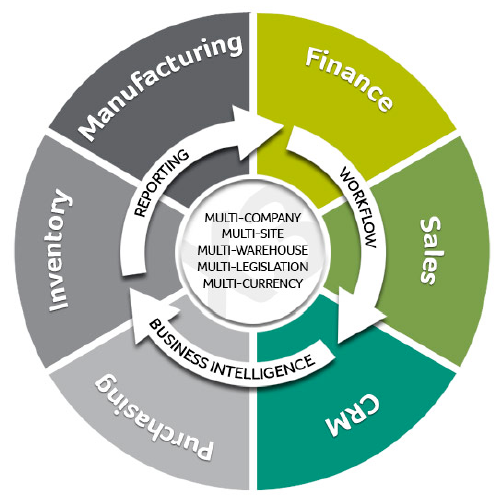 Identification and Traceability
Identification and Traceability
The nature of this industry calls for controlled processes to develop and manufacture products that meet the strictest quality standards. If a defect does occurs in product, immediate action needs to be taken to not only prevent consumers from serious health problems or death, but also to protect the viability of your business.
Creating and maintaining trace relationships between lots is arduous and time-consuming. Sage ERP X3 provides complete forward and backward lot traceability. A full audit trail and archive of historical transactions is maintained for multi-year periods. The fully integrated quality control process rigorously enforces inspections to assure item conformance to any required characteristics, operational tolerances or expected results.
About Us
Greytrix Africa is a leading Implementation, Reselling and Consulting partner for Sage Enterprise Management (Sage X3), Sage 300, Sage Enterprise Management HR (Sage X3 People) & Sage CRM based out of Nairobi, Kenya in East Africa region catering to Tanzania, Ethopia, Middle East, Australia and Asia businesses.
Greytrix Africa offers professional services such as Implementation & configuration, Business Process Analysis, Project Management, Customization, Integrations & Migrations, Technical support and Onsite resource augmentation along with enhancements within Sage Enterprise Management (Sage X3), Sage Enterprise Management HR (Sage X3 People), Sage CRM and Sage 300 (Accpac) across various industry verticals like Process Manufacturing (Food & Beverages, Chemical), Discrete manufacturing (Automotive, Textile & Apparel), Non Profit, Health-care Industry and Services Industry (Financial , Software & Engineering).
For more information on Sage Enterprise Management Consulting Services, please contact us at sales@greytrix.com.We will be glad to assist you.
